To protect the consumer, the FDA monitors domestic and foreign food businesses and the safety of the products they offer for sale on the US market. The data gathered from FDA inspections ensure a robust level of food safety protection. FDA is involved in various activities such as inspection, recall, sampling, seizure of food products including criminal prosecution of food companies.
The FDA’s New Era of Smarter Food Safety initiative, under the leadership of Frank Yiannas, Deputy Commissioner for Food Policy and Response, aims at curbing the number of foodborne illnesses and recalls in the US. This initiative relies on FSMA, the modernized food safety regulatory framework created by the FDA. FSMA rules such as the Preventive Controls for Human Food, the Foreign Supplier Verification Program or The Food Defense Rules are examples of regulations that food businesses including importers must abide under FSMA.
How FDA-Track (Food) Dashboards Are Helping Improve Food Safety
FDA-Track (Food) provides food safety compliance statistics for the development of preventive control plans for human food or food importation/supplier approval programs. The following portals are featured:
FSMA Dashboards – PC and cGMP measures
FDA is collaborating with the Centers for Disease Control and Prevention (CDC) to gauge the effect of the Current Good Manufacturing Practice, Hazard Analysis, and Risk-Based Preventive Controls for Human Food regulation (CGMP & PCHF regulation) on outbreaks of foodborne diseases in the United States.
Main Objectives:
- Increased Industry Compliance with Preventive Controls Regulations
- Fast and Effective Recalls
The dataset is downloadable for review and analysis. Select the PC and cGMP measures menu option.
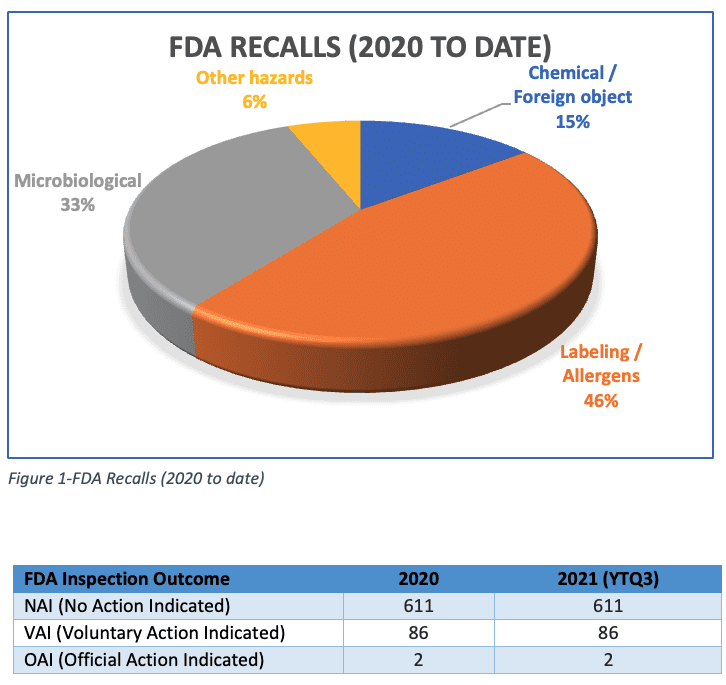
Note (Source: FDA, 2022):
No Action Indicated (NAI) which means no objectionable conditions or practices were found during the inspection (or the objectionable conditions found do not justify further regulatory action), Voluntary Action Indicated (VAI) which means objectionable conditions or practices were found but the agency is not prepared to take or recommend any administrative or regulatory action, or Official Action Indicated (OAI) which means regulatory and/or administrative actions will be recommended.
Reportable Food Registry Data Dashboard
The Reportable Food Registry (RFR or the Registry), established in 2007, is an electronic portal for industry to report food safety incidents when there is a “reasonable probability” that food will pose “serious adverse health consequences.” The registry helps the FDA better protect the consumer by trending food safety compliance data. The portal is used by the food manufacturing industry and public health officials.
The RFR dataset is available for download and contains 10 years of data from September 2009 to 2019. 28 commodities and 20 food safety hazards are tracked. The dashboard allows users to interact with the data points to gather more insights and to collect information based on selected foods, food safety hazards and time frames.
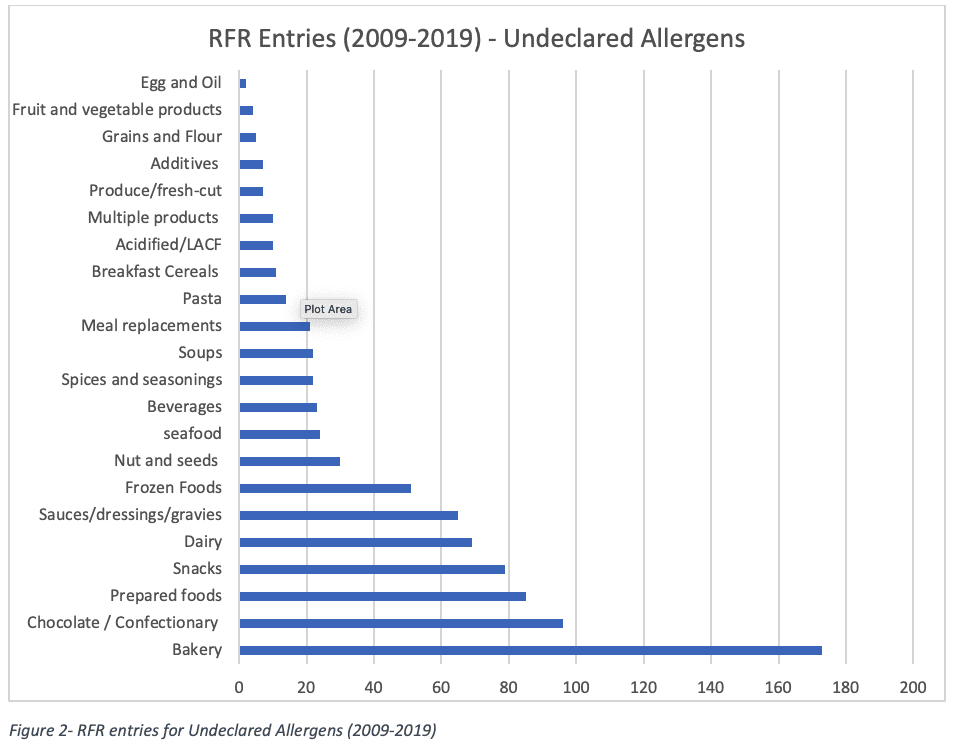
A Look at the RFR Data
For the period 2009-2019, the RFR has received 10,982 Total Reportable Entries. Undeclared Allergens represent 847 entries (40% of all entries), amongst which the baking industry, confectionary/chocolate, prepared and snack foods make the bulk of all acceptable submissions as shown below.
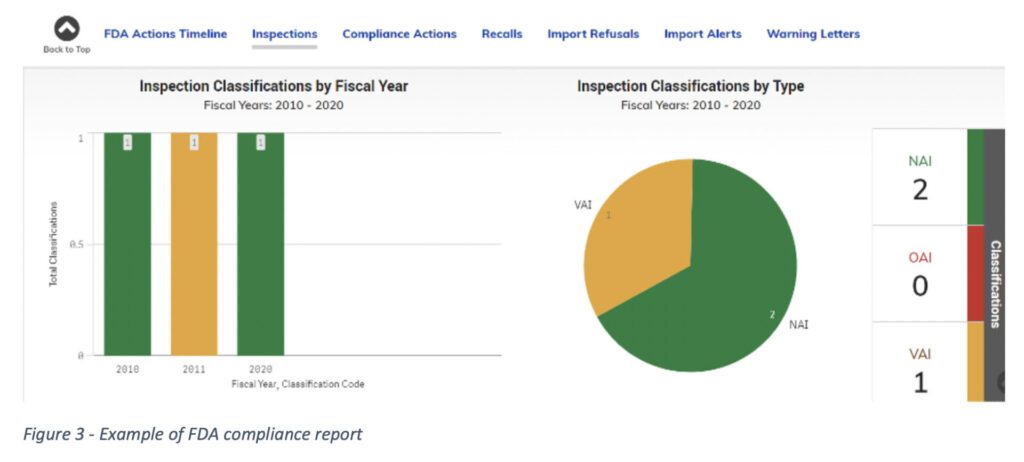
The information provided on the FDA Data Dashboard includes inspection and compliance data for FDA registered facilities. The information pertaining to domestic and foreign food businesses is updated weekly and includes final enforcement action detail. As per the FDA, “the Data Dashboard [was created] to increase transparency and accountability by displaying and allowing the analysis of public FDA data through easy to use, visually accessible, customizable, and understandable graphics.”
The database is searchable based on company name or Firm Establishment Identifier (FEI number). Different menu options are available including inspection information, compliance actions, recalls, imports summary, import refusals, and import entry.
You can subscribe to FDA-Track updates at this link.





