Corrective Action and Preventive Action (“CAPA” for short) has always been a best practice requirement in the manufacturing sector. In the food industry, corrective action may not have been as vigorously enforced as in other regulated industries.1 Borrowing from the tenets of ISO business standards, the most advanced food safety quality management systems such as SQF (Safe Quality Food) require robust CAPA programs.
At the core of CAPA is the concept of Root Cause Analysis (RCA). Root Cause Corrective Action is a diagnostic tool that is used to identify the factor(s) responsible for an operating problem so that an adequate control may be applied to avoid recurrence. In simpler terms, it is a way to pinpoint what went wrong and how to successfully fix it.
Who should perform the RCA?
Though it is possible for a sole SQF practitioner to conduct an RCA, it is preferable that the whole SQF team be involved. As with the development of a HACCP plan, a cross-functional team is recommended for analyzing the problem from difference angles. Some quality and food safety issues affecting food products are complex and require a thorough investigation. RCA is likely to be successful if a group that is familiar with the product, the process or the equipment tackles the issue.
CAPA Process Steps2
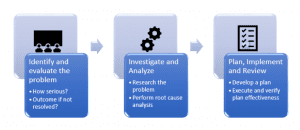
CAPA benefits
Implementing Corrective Actions is not only required by SQF version 8.0, but also mandated by FSMA (21 CFR § 117.150). Planning to implement detailed and systematic CAPA and RCA programs will go a long way towards resolving non-conformities and preventing recalls. In the context of SQF systems, it will also be a way to demonstrate continuous improvement while making internal audits more actionable.
Requirements of SQF Code, edition 8
2.5.3 Corrective and Preventative Action (Mandatory)
2.5.3.1 The responsibility and methods outlining how corrections and corrective actions are determined, implemented and verified, including the identification of the root cause and resolution of non-compliance of critical food safety limits, and deviations from food safety requirements, shall be documented and implemented.
2.5.3.2 Records of all investigation and resolution of non-conformities including their corrections and corrective action shall be maintained.
Tips on Conducting Effective RCA and Verification of Effectiveness
Using the scientific method can help you perform your Root Cause Analysis
Whether your CAPA team already employs the 5-WHY method or the fishbone diagram to perform RCA, it may also benefit from applying the principles of the scientific method.3 The six steps of this approach can be summarized as follows:
- Observe the process, collect data
- Analyze the data collected
- Form a hypothesis that extends beyond the available data – (example 1: product defect occurs regardless of what shift the product comes from, example 2: product non-conformity only occurs on line 2)
- Assume the hypothesis is true and test it (example 1: product manufactured on morning, afternoon and night shifts will be equally affected, example2: only line 2 products will be affected)
- Predict what the test results would be if the hypothesis is true (example 1: test results for morning, afternoon and night shift will be positive for test x, y and z, example2: test results for line 1 will be negative while test results for line 2 will be positive)
- Test the hypothesis (example 1 and 2: we will conduct test x, y, z to test this hypothesis and will analyze the data)
This process is helpful in identifying real sources of variation in a process that lead to non-conformities (such as equipment or work-force related non-conformities). You may repeat the process to continue the investigation until the real root cause of the problem is identified.
SMART objective concept applied to CAPA verification
The verification of effectiveness is often a missing step in implementing CAPA successfully. It consists of auditing the process to validate that the planned corrective actions have yielded the expected outcomes and that there were no unexpected negative consequences to implementing the CAPA plan. Knowing when to close an open Corrective Action Report is critical. Poor CAPA programs often result in recurring non-conformities and redundant corrective action plans. Applying the S.M.A.R.T method to the CAPA process can ensure that your corrective action plans are documented and effective.3 Practitioners are required to answer the following question: are the verification activities Specific, Measurable, Achievable, Relevant and Time bound? A well-documented verification of effectiveness is a way to demonstrate compliance with regulations, standards and business requirements.
References
1- SAI Global Course Manual – SQF Internal Auditing
2- Food Safety Tech- CAPA and Root Cause Analysis for the Food Industry
3- ASQ Quality progress magazine – March 2017





